HiToP® PNP is an advanced, patented medical device developed by gbo Medizintechnik AG (Germany), specifically designed for the treatment of Peripheral Polyneuropathy (PNP).
Using the patented HighTone Therapy® technology, HiToP® PNP helps reduce symptoms such as numbness, tingling, burning sensations, pain, and sensory disturbances in the feet and lower limbs—without medication and without known side effects.
No numbness – No paresthesia – Feel your feet again



The HighTone Therapy® technology is internationally patented and exclusively developed by gbo Germany, clearly differentiating HiToP® PNP from conventional electrotherapy systems such as TENS.
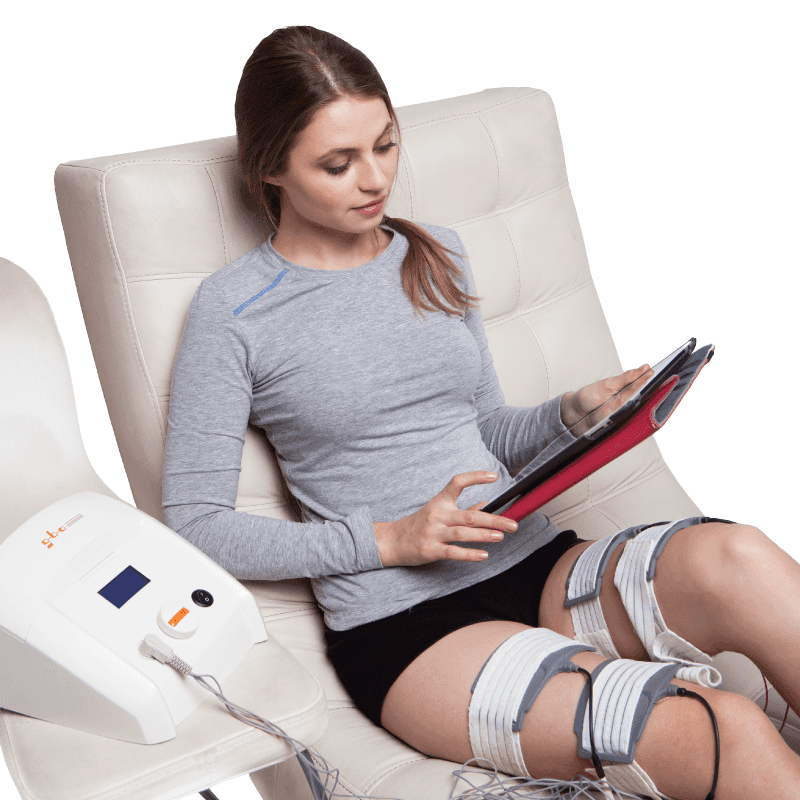
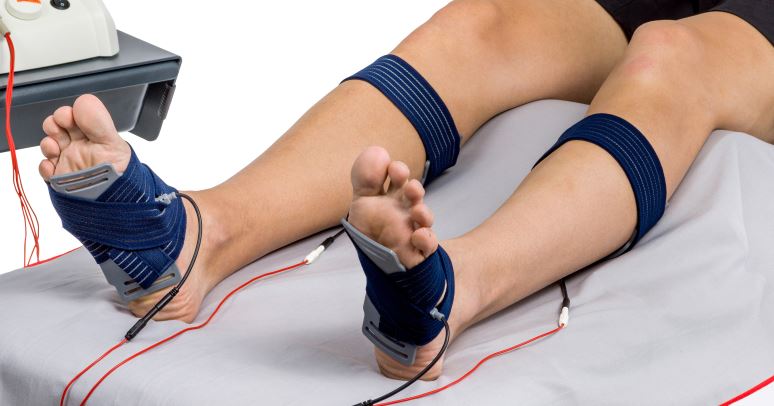
How HiToP® PNP Works
HiToP® PNP applies high-frequency electrical stimulation to muscles and peripheral nerves.
Unlike conventional low-frequency stimulation, HighTone Therapy® allows:
Deep and uniform nerve stimulation
Improved nerve signal transmission
Enhanced local blood circulation
Activation of cellular metabolism
This results in a significant reduction of neuropathic symptoms and a noticeable improvement in mobility and quality of life.
Key Benefits of HiToP® PNP
✔️ Patented HighTone Therapy® technology
✔️ Designed specifically for peripheral neuropathy (PNP)
✔️ Ideal for diabetic and non-diabetic neuropathy
✔️ Non-invasive and drug-free therapy
✔️ Safe and comfortable treatment sensation
✔️ Designed for home use (Home Use)
✔️ Medical-grade device – Made in Germany
Technical Specifications – HiToP® PNP
Specification | Details |
|---|---|
| Product Name | HiToP® PNP |
| Technology | Patented HighTone Therapy® |
| Channels | 1 channel |
| Display | LCD |
| Operating Frequency | 20 Hz |
| HighTone Frequency Range | 4,096 – 32,768 Hz |
| Modulation | 20 Hz |
| Power Supply | 100–240 V |
| Mains Frequency | 50–60 Hz |
| Max. Power Consumption | 50 VA |
| Protection Class | Class II (IEC 601-1) |
| Degree of Protection | BF (IEC 601-1) |
| Medical Device Class | IIa (according to MDD) |
| Dimensions (HxDxW) | 13.5 × 23 × 23 cm |
| Weight | 1.4 kg (without accessories) |
| Intended Use | Home Use |
| Warranty | 24 months |
| Manufacturer | gbo Medizintechnik AG – Germany |
Main Applications
Peripheral Polyneuropathy (PNP)
Diabetic neuropathy
Numbness and tingling of the feet
Burning sensations in the lower limbs
Chronic nerve pain
Sensory disorders caused by nerve damage

FAQ – Frequently Asked Questions
Is HiToP® PNP a patented technology?
Yes. HighTone Therapy® is a patented technology, exclusively developed and owned by gbo Medizintechnik AG (Germany).
Is the treatment painful?
No. Due to the high-frequency stimulation, treatment is very comfortable and usually perceived as pleasant by patients.
Can HiToP® PNP replace medication?
HiToP® PNP is a drug-free therapy option and may help reduce dependency on pain medication. Medical consultation is recommended for individual treatment plans.
How long does one treatment session take?
A typical session lasts approximately 30 minutes, depending on the therapy protocol.
Is HiToP® PNP safe for home use?
Yes. The device is specifically designed for Home Use and complies with European medical safety standards.
Who should not use this device?
Patients with pacemakers or implanted electronic devices should consult a physician before using HiToP® PNP.
Inquiry about the product:
You have a question about our product? Please feel free to write us a message:





Reviews
There are no reviews yet.